How GLP-1, GIP, and Glucagon Pathways Work in Metabolic Research
Introduction
Incretin hormones play a central role in metabolic research due to their influence on appetite signaling, insulin dynamics, nutrient handling, and energy balance. Modern GLP-1–based peptide research increasingly evaluates how multiple pathways interact — particularly GLP-1, GIP, and glucagon signaling — to shape experimental outcomes in controlled models.
In this article, we explain how GLP-1, GIP, and glucagon pathways function in metabolic research frameworks and why multi-agonist peptide designs are frequently explored.
Disclaimer: All compounds discussed are intended strictly for laboratory research use only. Not approved for human use.
Quick navigation: Jump to Pathways | Jump to Multi-Agonists | Jump to Comparison
What Are Incretin Hormones?
Incretins are gut-derived hormones released in response to nutrient intake. In research models, incretin signaling is commonly studied because it links nutrient sensing to endocrine responses and central appetite pathways. The most discussed incretin pathways include:
- GLP-1: appetite signaling, gastric emptying dynamics, glucose-responsive insulin pathways
- GIP: complementary incretin signaling often studied alongside insulin and adipose-related endpoints
- Glucagon: energy balance and substrate utilization pathways commonly included in advanced models
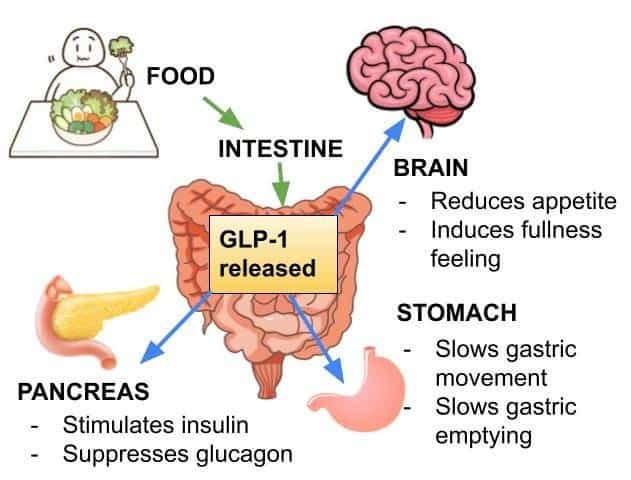
GLP-1 Pathway Explained
In experimental settings, GLP-1 signaling is commonly evaluated for its role in appetite-related neural pathways, gastric emptying effects, and glucose-dependent insulin dynamics. Research designs often track:
- Feeding behavior endpoints and intake patterns
- Glucose handling and insulin-response dynamics
- Downstream metabolic markers associated with incretin signaling
GIP Pathway Explained
GIP is often studied as a complementary incretin pathway. In dual-agonist research frameworks, GIP signaling is commonly explored for its interactions with GLP-1–mediated endpoints, especially in models where researchers track glucose handling and adipose-related markers.
- Dual incretin signaling designs (GLP-1 + GIP)
- Insulin dynamics under controlled nutrient conditions
- Metabolic endpoint expansion beyond GLP-1–only designs
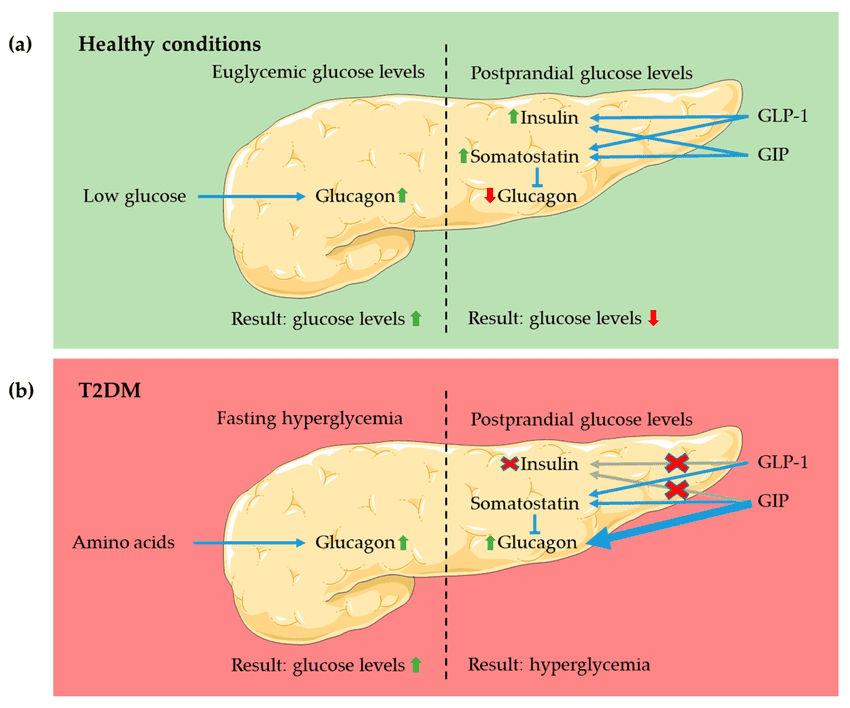
Glucagon Pathway Explained
Glucagon signaling is commonly included in more advanced metabolic research frameworks. In experimental designs, it is often associated with energy balance endpoints and substrate utilization parameters. Researchers may evaluate:
- Energy expenditure–related endpoints
- Lipid metabolism and substrate utilization markers
- Metabolic flexibility measures in controlled models
Why Multi-Agonist Peptides Are Studied
As research questions become more complex, some experimental designs explore multi-agonist approaches to evaluate broader pathway coverage. Compared to single-pathway signaling, multi-agonist designs can help researchers examine multiple endpoints within the same framework, depending on the model and controls used.
- Single-agonist: GLP-1–focused signaling frameworks
- Dual-agonist: combined GLP-1 + GIP endpoint expansion
- Triple-agonist: GLP-1 + GIP + glucagon pathways included in advanced designs
Mechanisms Overview Comparison
| Pathway | Primary Research Role | Common Endpoints |
|---|---|---|
| GLP-1 | Appetite + incretin signaling focus | Intake patterns, glucose handling, insulin dynamics |
| GIP | Complementary incretin signaling expansion | Glucose/insulin endpoints, adipose-related markers (model-dependent) |
| Glucagon | Energy balance and substrate utilization focus | Energy expenditure proxies, lipid metabolism markers, flexibility endpoints |
Comparing GLP-1 Research Peptides
When selecting a GLP-1–based research peptide, many researchers compare whether the design requires GLP-1–only signaling or expanded dual/triple pathway coverage.
Related: Semaglutide vs Tirzepatide vs Retatrutide (Comparison Guide)
Synergistic Research Stacking
- GLP-1 + NAD⁺: Often explored when metabolic signaling is paired with cellular energy and mitochondrial-related endpoints.
- GLP-1 + Glutathione: Commonly considered in oxidative stress and redox-balance research frameworks.
Best Practices for Metabolic Pathway Research
- Use clearly defined endpoints and consistent dietary controls
- Track pathway-relevant markers using standardized assay approaches
- Maintain consistent storage/handling to reduce degradation risk
- Confirm compound quality with COA and HPLC validation
- Ensure compliance with research-only regulations
Final Thoughts
GLP-1, GIP, and glucagon pathways each contribute distinct signaling roles within metabolic research frameworks. Understanding how these pathways intersect can help researchers structure controlled experimental designs and determine whether single-, dual-, or triple-agonist approaches best align with the endpoints being measured.
At ReviveLab, we supply COA-verified, research-grade peptides to support reproducible laboratory work across metabolic pathway research models.
All compounds are intended strictly for laboratory research use only. Not for human consumption.